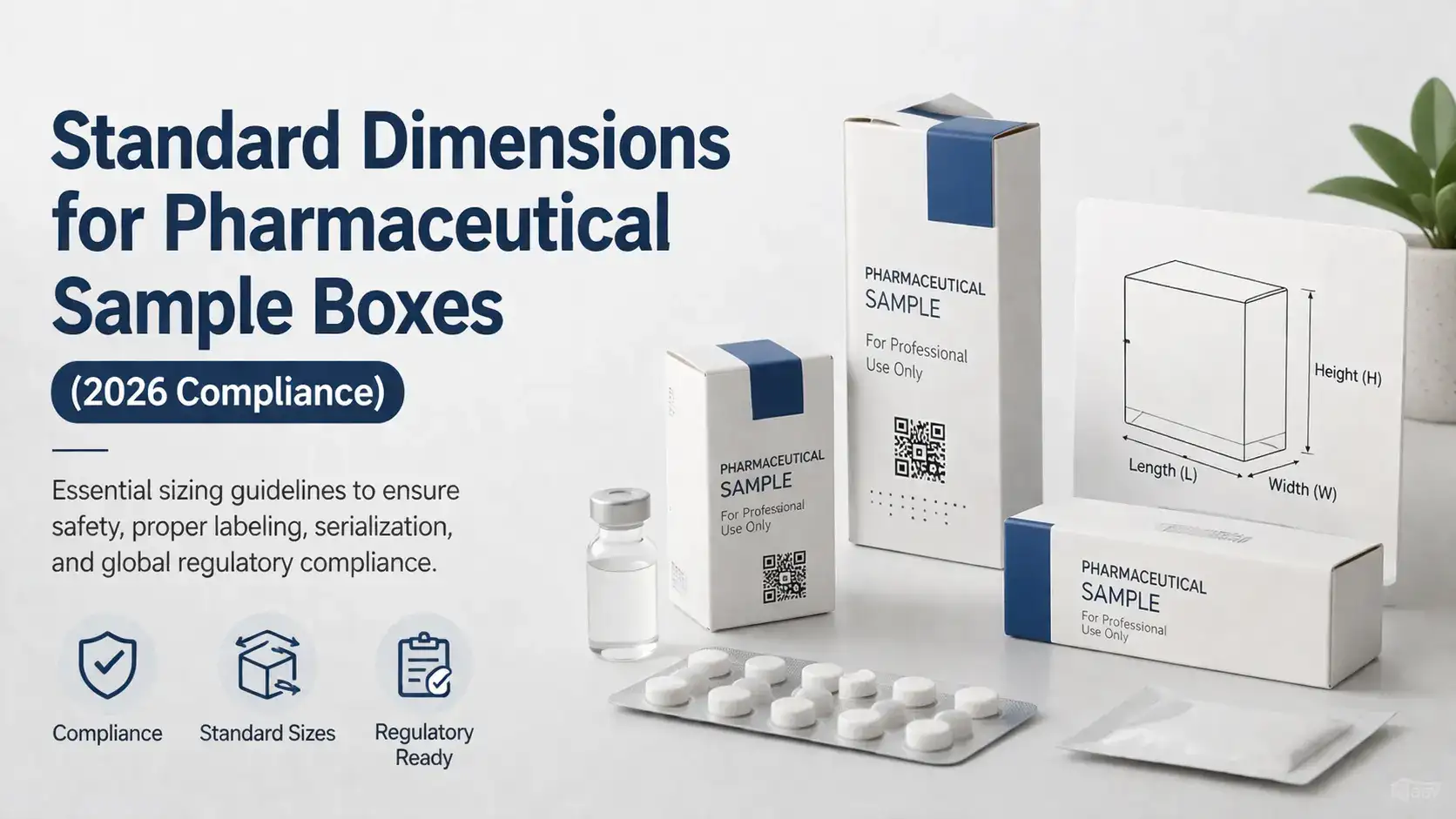
Pharmaceutical sample boxes are not simple packaging. They keep the product safe within. They contain very important data. They have to be very strict with the requirements. And in 2026, the requirements are more detailed than ever before. To manufacturers, packaging purchasing departments, and compliance departments, learning the standard size of pharmaceutical sample boxes is one facet of remaining on the right side of regulations.
Size may not be a big issue. In pharmaceutical packaging, size directly affects safety, standards, and functionality.
A small box will not carry the necessary information in labeling. An oversized box leads to unwanted movement of the product within it, increasing the risk of damage. The dimensions also influence how boxes are stored, transported, and displayed. In controlled markets, the dosage visibility will be associated with legally enforced packaging sizes, tamper-evidence, and child-resistance.
Providing inaccurate dimensions results not only in practical issues. It may lead to regulatory rejection, product recalls, and significant financial losses.
There is no international system that establishes the universal size of a pharmaceutical sample box. The sources of standards include a mixture of regional, international, and industry standards-setting organizations.
The Food and Drug Administration in the United States provides packaging mandates in the form of guidance documentation. In Europe, packaging standards are regulated by the European Medicines Agency and European directives on pharmaceuticals. Internationally, health authorities in most other countries follow the recommendations of the World Health Organization or use adapted versions of US or EU standards.
The introduction of recommendations to the 2026 compliance deal primarily addresses serialization, labeling space, and access for vulnerable groups such as older people and the visually impaired. Such developments directly affect the smaller sizes deemed acceptable by pharmaceutical sample boxes.
One size does not fit all pharmaceutical boxes. They are divided into general categories based on the products they hold. Each category includes dimensional specifications that are determined not only by regulatory requirements but also by industry standards.
Some of the most common pharmaceutical boxes are blister pack cartons. They carry blister strips that are push-through and hold pills or capsules. Small sample cartons within this group of standards generally measure about 55mm by 35mm by 10mm in length, width, and depth, respectively.
These dimensions fit standard blister strip sizes without taking away the space needed for the package insert and labeling. By 2026, revised EU serialization will require the carton to have a minimum flat surface area, with a data matrix code of at least 10mm x 10mm encoded. This has also forced up the minimum width dimensions in some markets.

Sample boxes containing small vials, ampoules, or small glass or plastic bottles are subject to different dimensional requirements. These are squarer in cross-section and taller. Common sizes are between 40mm and 80mm wide, and 60mm to 150mm high, depending on the size of the container housed.
The inner dimensions should provide a tight fit with the main container and sufficient cushioning to prevent any movement. Excessive internal space is a compliance problem in certain markets due to the implications of false packaging volumes.
The boxes of sachets, powder packets, or single-dose stick packs are more likely to be flatter and wider. Such sizes allow several sachets to be placed in the box without folding or bending.
Injectable product sample boxes do not pose a general dimensional requirement due to the characteristics of the main container. The syringe will need to be safely placed in these boxes in such a position. They are commonly tray-and-lid shapes. The syringe is fixed in internal molded trays or inserts to prevent movement during transportation, ensuring injectable pharma packaging standards. Take a look at different varieties of lid packaging boxes to determine if they are the right fit for your product or not.

Space requirements are considered one of the primary contributors to the minimum box dimensions in 2026. Regulatory authorities require that some information be on the exterior of each pharmaceutical box. This entails the product name, active ingredient, and strength, batch number, expiry date, manufacturer details, storing conditions, serialization code, and, in most countries, Braille text.
The European Falsified Medicines Directive, as well as the regulations of other jurisdictions, state that the serialization data matrix code should be scannable with a clear zone. This, in itself, demands a minimum panel size that many older and smaller sample box designs cannot accommodate.
Another dimension of consideration is Braille needs. Braille text must have a certain dot size and distance. The surface on which the Braille can be written should be flat and large enough. This usually requires the highest or lowest panel of the box to have a minimum area. In reality, boxes smaller than 35mm in width struggle to display readable Braille text.
Labeling with a small font size reduces the surface area required. The key information in the EU has to be at least 7-point font. In the US, legibility standards set realistic minimums that affect panel size.

Pharmaceutical lid packaging boxes are almost universally made from folding boxboard. This board thickness influences the size of the outside of the box made compared to the inside.
Pharmaceutical carton weights standard boards are between 230gsm and 350gsm. Board thickness at such weights is usually 0.3mm to 0.5mm. The case of a six-sided box is to have the outer dimensions increased by 0.6mm to 1mm relative to the inner dimensions at each side. This might appear insignificant, but it is important when designing boxes that fit secondary packaging or run on automatic lines for filling and cartoning.
To comply with 2026, along with any similar packaging specifications, it is essential to indicate whether the dimensions are inner or outer explicitly. The two are often confused, hence introducing errors when submitting a regulatory submission.
There are additional dimensional constraints of child-resistant packaging standards. The Poison Prevention Packaging Act in the US specifies requirements for certain types of pharmaceuticals. EN 14375 and EN 862 are the EU's child-resistant requirements for reclosable and non-reclosable formats, respectively.
Child-resistant designs, such as push-and-turn panels, inbuilt tear strips, or interlocking closures, add more material and also a little more size to the box (not structural integrity) to fit the mechanism. Meanwhile, packaging that is senior-friendly, introduced in various markets, must be easy to open without an awkward or straining effort. The two requirements are sometimes contradictory. The challenge for pharmaceutical packaging designers remains to balance them while staying within the overall dimension parameters in 2026.
In 2026, serialization compliance mandates assigning a distinct identifier to each pharmaceutical sample box. This identifier is coded as a 2D data matrix. The code should conform to the ISO 15415 print quality standards.
The artistic code files submitted for regulatory review should indicate the precise location of the serialization code on the box plan. The code should not be located on a fold line, a perforated field, or a covering that a label, sticker, or similar will cover. These placement rules have a direct impact on the design of a box and an indirect effect on the available surface area for other labeling elements.
The Pharmaceutical sample box sizes in 2026 will be determined through a mix of product type, regulatory labeling, and serialization standards, as well as accessibility concerns. Knowing these aspects and using them properly in the design phase will avoid costly redesign work and ensure that products remain compliant across all markets they penetrate.